Abstract
Objective: In this study, we investigated the initial serum immunoglobulin and lymphocyte subset levels of children with immune thrombocytopenia (ITP), and their association with treatment response.
Methods: Thirty children with ITP were retrospectively analyzed. Immunoglobulin isotypes, IgG subtypes, and lymphocyte subset levels in the patients were compared with those of the age- and sex-matched control group and age-appropriate reference values. In addition, we investigated the relationship between immunological parameters and treatment responses.
Results: There was no statistically significant difference between the patient and control groups regarding immunoglobulin isotype and IgG subtype levels. However, IgG levels were below normal limits in 10 (33.3%) patients, and IgA and IgG2 levels were low in 2 patients with normal IgG levels. Younger age and low IgG level at diagnosis were associated with an increased treatment response on day 7.
Conclusion: Serum immunoglobulin levels at the time of diagnosis can help predict treatment response in the early period and detect subclinical immunodeficiency in children with acute ITP.
Keywords: childhood, acute immune thrombocytopenia, immunodeficiency
INTRODUCTION
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by isolated thrombocytopenia and is the most common cause of thrombocytopenia in children. The incidence of ITP in children ranges from 1.9 to 9.5/100,000 children annually. A seasonal fluctuation is observed, peaking in late winter and early spring.1-5 Immune thrombocytopenia can be divided into three classes according to the duration of thrombocytopenia: It is called newly diagnosed ITP in the first three months after diagnosis, persistent ITP between 3 and 12 months after diagnosis, and chronic ITP if it lasts longer than 12 months.6
Primary immunodeficiencies are a group of diseases comprising over 500 diseases.7 They can be seen at any age, but are much more common in childhood. There are delays of up to five years in diagnosing primary immunodeficiency patients, which may result in irreversible complications, especially bronchiectasis, after recurrent respiratory tract infections.8-10
In patients with primary immunodeficiency, susceptibility to autoimmune and infectious diseases increases. Common pathophysiological and genetic risk factors have been described for autoimmunity and primary immunodeficiency. Selective immunoglobulin A (IgA) deficiency is the most common primary immunodeficiency disease. The frequency of autoimmune diseases, including ITP, is higher in patients with selective IgA deficiency than in the general population.8,11,12 In addition, autoimmune disease was found in approximately 20-30% of patients with common variable immunodeficiency, the second most common disease among all primary immunodeficiencies, and ITP has been detected in 10-12% of them.8,13,14
Autoimmunity may be the first manifestation in patients with primary immunodeficiency, and thrombocytopenia is the most common hematological finding.8,15,16 Therefore, children with immune thrombocytopenia should be examined, considering that primary immunodeficiency may also be present.16,17
All these findings indicate that there may be some changes in the immune systems of children with ITP. We hypothesize that these changes, which may be related to disease onset, may also provide insight into disease prognosis. In this study, we investigated two issues: 1-Are the serum immunoglobulin levels and lymphocyte subsets at the time of diagnosis different from those of healthy children in children with acute ITP? 2- Is there a relationship between serum immunoglobulin and lymphocyte subset levels and treatment response?
MATERIALS AND METHODS
This retrospective study was carried out in the Sakarya University University Faculty of Medicine, Department of Pediatric Hematology and Oncology, and approved by the Ethics Committee of the Sakarya University Faculty of Medicine (March 30, 2021; E-71522473-050.01.04-21476-233). Patients under 18 years of age who were followed up with a diagnosis of immune thrombocytopenia in the Department of Pediatric Hematology and Oncology between 2018 and 2021 were included in the study. The diagnosis of immune thrombocytopenia was made based on the history, physical examination, and laboratory data. Children with platelet count <100,000/µL, average white blood cell count and hemoglobin values, and no findings suggestive of another cause for thrombocytopenia (a sign of hemolysis or blast on peripheral smear; positive direct Coombs test or antinuclear antibody; lymph node, spleen or liver enlargement; joint anomalies and presence of hyper or hypopigmented skin lesions; family or patient history of abnormal bleeding) as a result of examination and laboratory tests were defined as ITP. Patients with thrombocytopenia lasting less than one year were accepted as acute or persistent immune thrombocytopenia and included in the study. However, patients who had been symptomatic for longer than 1 year were considered to have chronic immune thrombocytopenia and were excluded from the study.
Demographics of the patients, laboratory test results at admission (platelet, white blood cell, neutrophil, and lymphocyte counts in peripheral blood; Immunoglobulin isotype, IgG subtype, complement C3, C4, and lymphocyte subset levels), treatment modalities, and platelet counts measured in the control periods (7, 15, and 30. days; 2, 3 and 6. months) were recorded. An increase in the platelet count to >30,000/µL was considered a positive response to treatment (responder), and <30,000/µL was considered a negative response (non-responder).18 Patients were divided into two groups, “low” and “normal,” according to the age-appropriate reference ranges for lymphocyte subsets19, immunoglobulin isotypes20, IgG subtypes21 and complement22 levels. We evaluated values below the lower limit of 95% confidence interval (<-1.96 SD) as low.
The control group was selected from children referred to the Pediatric Immunology Outpatient Clinics at Sakarya University and were deemed healthy. In this study, only immunoglobulin isotype and IgG subtype levels were compared between the healthy control and patient groups.
Statistical analyses
The numerical variables used in the study were evaluated for normality using the Kolmogorov-Smirnov test. All variables except some had a normal distribution. Logarithmic transformation was performed on variables that did not follow a normal distribution, and the transformed data showed a normal distribution. All numerical variables were presented as mean ± standard deviation. Categorical variables were expressed as numbers and percentages. Numerical variables were compared using the independent samples t-test. Categorical variables were compared using the chi-square test. Repeated-measures ANOVA was used to assess whether platelet counts changed during the control periods. Logistic regression analysis and the Backward Wald elimination model were applied to determine the factors affecting the treatment response rate in the control periods. Type I error (α) was predicted as 0.05 in the study, and it was considered statistically significant when the p-values were below 0.05. All statistical analyses were performed using software (IBM SPSS Statistics, Version 23.0, Armonk, NY: IBM Corp.).
RESULTS
The study was conducted on 30 children with ITP (70.03±57.07 months of age; 13 males, 17 females) and 30 age and sex-matched healthy controls (70.08±57.3 months of age; 14 males, 16 females).
There was no statistically significant difference between the patient and control groups regarding immunoglobulin isotype and IgG subtype levels. However, in 10 (33.3%) patients with a normal CD3+ cell ratio, IgG levels were below normal limits, and this was accompanied by at least one decrease in IgA or IgM according to the age-related reference values of serum immunoglobulin levels in healthy Turkish children. These patients were considered to have a suspected diagnosis of common variable immune deficiency. In addition, IgA and IgG2 levels were low in two of 14 patients with normal IgG levels but low levels of one IgG subtype. These patients may also have selective IgA deficiency. However, the diagnosis could not be confirmed in both groups.
Children with ITP were divided into “low” and “normal” subgroups according to immunoglobulin isotype and IgG subtype levels. At least one immunoglobulin isotype or subtype level was lower than the age-appropriate reference values, except in one patient. While serum IgA levels were below normal in 26.7% of patients, IgM levels were low in 60% of patients (Table 1).
| Table 1. Immunoglobulin isotypes, IgG subtypes, and lymphocyte subset levels of the patients according to the age-appropriate reference range | |||||
|---|---|---|---|---|---|
| CD3+: cluster of differentiation 3 positive; CD4+: cluster of differentiation 4 positive; CD8+: cluster of differentiation 8 positive; CD19+: cluster of differentiation 19 positive; IgA: immunoglobulin A; IgG: immunoglobulin G; IgG1: immunoglobulin G1; IgG2: immunoglobulin G2; IgG3: immunoglobulin G3; IgG4: immunoglobulin G4; IgM: immunoglobulin M; NK: natural killer. | |||||
| Immunoglobulin level |
|
|
|
||
| IgG | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgA | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgM | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgG1 | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgG2 | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgG3 | Low |
|
|
|
|
| Normal |
|
|
|
||
| IgG4 | Low |
|
|||
| Normal |
|
||||
Regarding lymphocyte subset levels, at least one subset value was below the age-appropriate reference range except in one patient. The percentage of CD3+CD8+ cells was lower than normal in 3.3% of patients, whereas 40% of patients had a lower-than-normal CD19+ cell population (Table 1).
There was a statistically significant difference (p<0.001) between the platelet counts measured at the time of the diagnosis and on the 7th day, 15th day, 30th day, second month, third month, and sixth month after the diagnosis (Figure 1). As a result of pairwise comparisons, it was determined that the mean platelet value at the time of diagnosis was statistically significantly lower than all other measurements except the 7th day; and the 7th, 30th day, and 2nd month values were lower than the 3rd and 6th months.
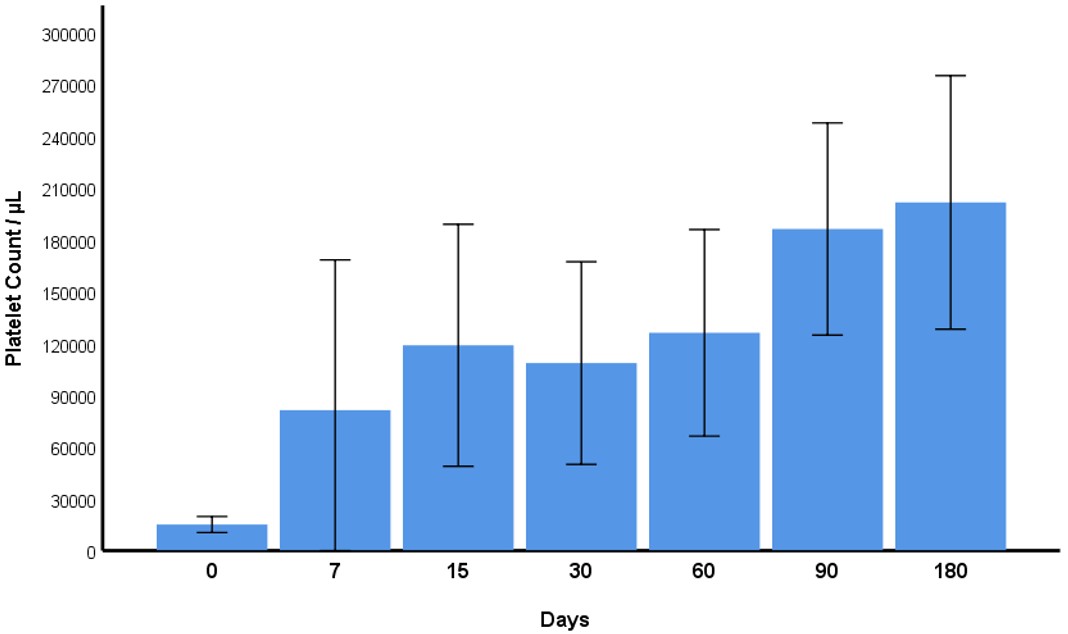
Sixty-seven percent (67%), 68%, and 76% of patients responded to treatment (platelet count >30,000/µL) at the 7th, 15th, and 30th days, respectively, after treatment.
Ten (33.3%) children were treated with IVIG, eight (26.7%) with corticosteroids, and seven (23.3%) with sequential treatment (IVIG + corticosteroid); five (16.7%) were followed without treatment. The response rates of the treatment groups were similar (Table 2).
|
* Fisher’s Exact Test was used for statistics. IVIG: intravenous immunoglobulin; W/O: without. |
||||||
| Table 2. Comparison of the response rates of the treatment groups | ||||||
| Response |
|
|
|
|
|
|
| 7. day | Negative |
|
|
|
|
|
| Positive |
|
|
|
|
||
| 15. day | Negative |
|
|
|
|
|
| Positive |
|
|
|
|
||
| 30. day | Negative |
|
|
|
|
|
| Positive |
|
|
|
|
||
The age of the patients who responded to treatment on day seven was significantly lower than that of those who did not respond. However, there was no difference between the groups on days 15 and 30. There was no difference between the groups regarding white blood cell, neutrophil, and lymphocyte counts on days seven, 15, and 30.
Patients who did not respond to treatment on day seven had significantly higher IgG and IgA levels at diagnosis (973.6±311.8 mg/dL and 120.5±80.4 mg/dL) than responders (752.6±248.6 mg/dL and 70.3±49.7 mg/dL); however, IgM, IgG subtypes, and complement levels were similar in both groups (Table 3). There was no difference between the two groups in immunoglobulin isotypes, IgG subtypes, or complement levels compared with responses at 15 and 30 days.
In cases with low IgG levels at diagnosis, the positive treatment response rate was significantly higher on day 7. There was no difference between patients with low IgA, IgM, and IgG subtype levels and those with normal levels. There was no difference in response rates between children with low and normal immunoglobulin levels on days 15 and 30 (Table 4).
| IgA: immunoglobulin A; IgG: immunoglobulin G; IgG1: immunoglobulin G1; IgG2: immunoglobulin G2; IgG3: immunoglobulin G3; IgG4: immunoglobulin G4; IgM: immunoglobulin M. | ||||
| Table 4. Comparison of treatment responses on day 7 according to immunoglobulin levels at diagnosis | ||||
|
|
|
|
||
| IgG | Low |
|
|
|
| Normal |
|
|
||
| IgA | Low |
|
|
|
| Normal |
|
|
||
| IgM | Low |
|
|
|
| Normal |
|
|
||
| IgG1 | Low |
|
|
|
| Normal |
|
|
||
| IgG2 | Low |
|
|
|
| Normal |
|
|
||
| IgG3 | Low |
|
|
|
| Normal |
|
|
||
| IgG4 | Low |
|
|
|
| Normal |
|
|
||
The relationship between lymphocyte subsets and treatment responses was also examined. The lymphocyte subset levels of the patients who responded and did not respond to treatment on the 7th, 15th, and 30th days were not different. Similarly, patients with low and normal lymphocyte subset levels had similar response rates on days seven, 15, and 30.
In logistic regression analysis, lower patient age and IgG level were associated with an increased treatment response rate on day 7 (Table 5). However, no factors affected the treatment response rate on day 15 and day 30.
DISCUSSION
Almost all children with acute ITP had either low serum IgG or low lymphocyte subset levels. In only one patient, both immunoglobulin levels and lymphocyte subset ratios were within the normal range. The response rates for patients with low IgG levels at diagnosis were significantly higher on the seventh day of treatment. In logistic regression analysis, a low IgG level was associated with an increased treatment response rate on day 7.
Rahiminjad et al.23 evaluated 36 patients with ITP, aged 3-51, and found no significant differences in immunoglobulin isotype and IgG subtype levels between the patient and control groups. However, they reported selective IgA deficiency in two (5.5%) patients and IgG subtype deficiency in four (11.1%) patients. In the present study, all three immunoglobulin isotype levels were normal in seven (23.3%) patients. However, one, two, and three different immunoglobulin isotype levels were below the reference values in 40%, 20%, and 16.7% of the patients, respectively. Ten (33.3%) patients had low IgG levels accompanied by low levels of at least one of the IgA and IgM. However, the CD3-positive cell ratios were normal. Although common variable immunodeficiency was suspected in these ten patients, the diagnosis could not be confirmed.
Serum immunoglobulin levels are among the most important screening tests for diagnosing primary and secondary immunodeficiencies. Common variable immunodeficiency progresses with low serum IgA or IgM levels in addition to low IgG.8 The frequency of autoantibodies increases in patients with both isolated and combined IgA and IgG2 deficiency.24 In this study, of the 17 patients with normal IgG isotype levels, 14 had a decrease in any of the IgG subtypes. IgA and IgG2 levels were low in 2 of these 14 patients. We thought these two children with normal IgG isotype levels might have selective IgA deficiency. Twenty percent of the healthy population may have low IgG subtypes and progress without clinical findings. These cases do not require treatment and should not be evaluated for immunodeficiency unless clinical findings are present.23
In a study examining lymphocyte distribution in the bone marrow of children with acute ITP, an increase in B cell precursor markers, such as CD10, CD19, and CD20, and a significant decrease in T cell markers (CD2, CD3, CD5, and CD7) were observed.25 Zahran et al.26 reported that the peripheral blood CD8+ and CD19+ cell ratios were significantly higher than the healthy group, and the CD4+ cell ratio was significantly lower in 40 children with acute ITP. In addition, there are differences in CD8+ and CD4+ T lymphocyte subpopulations. Central memory T (TCM) and CD8+ Naïve T cells are lower, and CD8+ T effector memory RA cells are higher; CD4+ TCM cells are lower; however, CD4+ T effector memory is higher in children with ITP than in controls.27 Unlike the above studies, we did not compare the patient results with the control group. We only determined those that were low or high according to the reference values. In the present study, the rate of CD19+ cells was below the reference range in 40% of the patients, and the percentages of CD8+ and CD4+ cells were mainly within the normal range. Two children with a low CD19+ cell percentage were accompanied by a low T cell (CD3+ in one patient, CD3+, CD4+ in another) and low immunoglobulin levels. Although these two patients had no history of recurrent severe infections, they were referred for evaluation for combined immunodeficiency. As a result, 10 (33.3%) patients were referred to the Immunology and Allergy Clinic with suspicion of common variable immunodeficiency, two (6.6%) patients with suspicion of combined immunodeficiency, and two patients with suspicion of IgA - IgG subtype deficiency.
We found that patients who responded to treatment on day seven were significantly younger, and in the logistic regression analysis, increasing age was associated with a decreased response rate to treatment. However, this effect was not observed on days 15 and 30. Additionally, during any of the control periods, there was no difference in white blood cell, neutrophil, or lymphocyte counts at the time of diagnosis between the treatment-responsive and non-responsive groups, nor in gender. In a multicenter cohort study involving 705 pediatric patients, remission rates at 12 and 24 months increased with decreasing age.28 Ahmed et al.29 reported that children’s age was not correlated with remission. Although white blood cell and lymphocyte counts in peripheral blood have been reported not to influence treatment success in adults with ITP, no study in children has demonstrated an association between these parameters and treatment response.30
Selective IgA deficiency was defined in 1% of the patients with ITP and common variable immunodeficiency in 1% of the patients, and it was found that elevated serum IgA and a decrease in serum IgM levels were associated with treatment-resistant ITP.31 However, this association was mostly demonstrated in patients aged 65 and over, and no relationship was found between serum immunoglobulin levels and the course of ITP in children under 18 years of age. Other studies have shown that high IgA and low IgM levels are associated with lower treatment success.32 In both studies mentioned above, treatment response was evaluated cumulatively, and no comparison was made in terms of responses at different periods, as we did. We detected an inverse relationship between immunoglobulin G and A levels at diagnosis and treatment response on day seven. However, there were no significant differences in IgM, IgG subtypes, and lymphocyte subset levels between the groups that responded and those that did not respond to treatment on days 7, 15, and 30. In addition, we did not find a significant difference between groups in treatment responses of patients with normal and high IgA levels during the control periods.
Autoantibodies against platelets are observed in approximately two-thirds of patients with ITP, most commonly IgG.3,33-36 In patients with ITP and high serum IgG levels, platelet surface IgG is also elevated, and the presence of high IgG on the platelet surface shortens platelet survival.37 In the light of the above studies, it can be speculated that the patients with low serum IgG levels in the current study also had low antiplatelet antibody levels and therefore had better responses on day seven.
The literature has reported that many changes in the T cell line play a role in the pathogenesis of ITP.38,39 In addition, regulatory T cells (Tregs) are lower in patients with ITP than in the healthy population, and there is a negative correlation with treatment response.26,40-42 Zhao et al.43 reported that adults with ITP who have higher levels of CD8+ cells or lower levels of CD4+/CD8+ cell ratio responded poorly to first-line steroid and IVIG treatments. However, Zahran et al.26 found that children with a high cytotoxic CD8+ T cell population at baseline had a higher recovery rate within the first three months. This study did not find a relationship between lymphocyte subset proportions measured at diagnosis and treatment response within the first 30 days.
A limitation of our study is that the patients’ serum immunoglobulin isotype and IgG subtype levels, and lymphocyte subgroup ratios, were evaluated only at the time of diagnosis. Since these measurements were not repeated, the diagnosis of immunodeficiency could not be confirmed, and the patients were referred to the Pediatric Immunology and Allergy Clinic for evaluation. In addition, the small sample size and the lack of evaluation of the treatment effects on clinical outcomes are other limitations of the study. In conclusion, serum immunoglobulin levels at the time of diagnosis can help predict treatment response in the early period and detect subclinical immunodeficiency in children with acute ITP.
Ethical approval
This study has been approved by the Ethics Committee of Sakarya University Faculty of Medicine (approval date 30.03.2021, number E-71522473-050.01.04-21476-233). Written informed consent was obtained from the participants.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Cooper N, Ghanima W. Immune thrombocytopenia. N Engl J Med. 2019;381:945-55. https://doi.org/10.1056/NEJMcp1810479
- Friedman JN, Beck CE. Diagnosis and management of typical, newly diagnosed primary immune thrombocytopenia (ITP) of childhood. Paediatr Child Health. 2019;24:54-5. https://doi.org/10.1093/pch/pxy197
- D’Orazio JA, Neely J, Farhoudi N. ITP in children: pathophysiology and current treatment approaches. J Pediatr Hematol Oncol. 2013;35:1-13. https://doi.org/10.1097/MPH.0b013e318271f457
- Shaw J, Kilpatrick K, Eisen M, Tarantino M. The incidence and clinical burden of immune thrombocytopenia in pediatric patients in the United States. Platelets. 2020;31:307-14. https://doi.org/10.1080/09537104.2019.1635687
- Terrell DR, Beebe LA, Vesely SK, Neas BR, Segal JB, George JN. The incidence of immune thrombocytopenic purpura in children and adults: a critical review of published reports. Am J Hematol. 2010;85:174-80. https://doi.org/10.1002/ajh.21616
- Rodeghiero F, Stasi R, Gernsheimer T, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113:2386-93. https://doi.org/10.1182/blood-2008-07-162503
- Poli MC, Aksentijevich I, Bousfiha AA, et al. Human inborn errors of immunity: 2024 update on the classification from the International Union of Immunological Societies Expert Committee. J Hum Immun. 2025;1:e20250003. https://doi.org/10.70962/jhi.20250003
- Amaya-Uribe L, Rojas M, Azizi G, Anaya JM, Gershwin ME. Primary immunodeficiency and autoimmunity: a comprehensive review. J Autoimmun. 2019;99:52-72. https://doi.org/10.1016/j.jaut.2019.01.011
- Joshi AY, Iyer VN, Hagan JB, St Sauver JL, Boyce TG. Incidence and temporal trends of primary immunodeficiency: a population-based cohort study. Mayo Clin Proc. 2009;84:16-22. https://doi.org/10.4065/84.1.16
- Panigrahi MK. Common variable immunodeficiency disorder - an uncommon cause for bronchiectasis. Lung India. 2014;31:394-6. https://doi.org/10.4103/0970-2113.142138
- Odineal DD, Gershwin ME. The epidemiology and clinical manifestations of autoimmunity in selective IgA deficiency. Clin Rev Allergy Immunol. 2020;58:107-33. https://doi.org/10.1007/s12016-019-08756-7
- Swain S, Selmi C, Gershwin ME, Teuber SS. The clinical implications of selective IgA deficiency. J Transl Autoimmun. 2019;2:100025. https://doi.org/10.1016/j.jtauto.2019.100025
- Cunningham-Rundles C. Autoimmune manifestations in common variable immunodeficiency. J Clin Immunol. 2008;28(Suppl 1):S42-5. https://doi.org/10.1007/s10875-008-9182-7
- Salzer U, Warnatz K, Peter HH. Common variable immunodeficiency: an update. Arthritis Res Ther. 2012;14:223. https://doi.org/10.1186/ar4032
- Abolhassani H, Amirkashani D, Parvaneh N, et al. Autoimmune phenotype in patients with common variable immunodeficiency. J Investig Allergol Clin Immunol. 2013;23:323-9.
- Westermann-Clark E, Meehan CA, Meyer AK, et al. Primary immunodeficiency in children with autoimmune cytopenias: retrospective 154-patient cohort. Front Immunol. 2021;12:649182. https://doi.org/10.3389/fimmu.2021.649182
- Bonilla FA, Khan DA, Ballas ZK, et al. Practice parameter for the diagnosis and management of primary immunodeficiency. J Allergy Clin Immunol. 2015;136:1186-205.e1-78. https://doi.org/10.1016/j.jaci.2015.04.049
- Neunert C, Lim W, Crowther M, et al. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011;117:4190-207. https://doi.org/10.1182/blood-2010-08-302984
- Comans-Bitter WM, de Groot R, van den Beemd R, et al. Immunophenotyping of blood lymphocytes in childhood. Reference values for lymphocyte subpopulations. J Pediatr. 1997;130:388-93. https://doi.org/10.1016/s0022-3476(97)70200-2
- Bayram RO, Özdemir H, Emsen A, Türk Dağı H, Artaç H. Reference ranges for serum immunoglobulin (IgG, IgA, and IgM) and IgG subclass levels in healthy children. Turk J Med Sci. 2019;49:497-505. https://doi.org/10.3906/sag-1807-282
- Aksu G, Genel F, Koturoğlu G, Kurugöl Z, Kütükçüler N. Serum immunoglobulin (IgG, IgM, IgA) and IgG subclass concentrations in healthy children: a study using nephelometric technique. Turk J Pediatr. 2006;48:19-24.
- Garcia-Prat M, Vila-Pijoan G, Martos Gutierrez S, et al. Age-specific pediatric reference ranges for immunoglobulins and complement proteins on the Optilite™ automated turbidimetric analyzer. J Clin Lab Anal. 2018;32:e22420. https://doi.org/10.1002/jcla.22420
- Rahiminejad MS, Mirmohammad Sadeghi M, Mohammadinejad P, et al. Evaluation of humoral immune function in patients with chronic idiopathic thrombocytopenic purpura. Iran J Allergy Asthma Immunol. 2013;12:50-6.
- Jiménez A, Alvarez-Doforno R, García Rodríguez MC, Ferreira A, López-Trascasa M, Fontán G. Autoantibodies in patients with IgA and IgG2 deficiencies. APMIS. 1991;99:327-32. https://doi.org/10.1111/j.1699-0463.1991.tb05157.x
- Alavi S, Aryan Z, Ghazizadeh F, Arabi N, Nikougoftar M, Ebadi M. The immunophenotype of bone marrow lymphocytes in children with immune thrombocytopenic purpura. Pediatr Hematol Oncol. 2014;31:548-54. https://doi.org/10.3109/08880018.2014.927944
- Zahran AM, Elsayh KI. CD4+ CD25+High Foxp3+ regulatory T cells, B lymphocytes, and T lymphocytes in patients with acute ITP in Assiut Children Hospital. Clin Appl Thromb Hemost. 2014;20:61-7. https://doi.org/10.1177/1076029612454937
- Zahran AM, El-Badawy OH, Mahran H, et al. Detection and characterization of autoreactive memory stem T-cells in children with acute immune thrombocytopenia. Clin Exp Med. 2024;24:158. https://doi.org/10.1007/s10238-024-01386-0
- Bennett CM, Neunert C, Grace RF, et al. Predictors of remission in children with newly diagnosed immune thrombocytopenia: data from the Intercontinental Cooperative ITP Study Group Registry II participants. Pediatr Blood Cancer. 2018;65:10.1002/pbc.26736. https://doi.org/10.1002/pbc.26736
- Ahmed S, Siddiqui AK, Shahid RK, Kimpo M, Sison CP, Hoffman MA. Prognostic variables in newly diagnosed childhood immune thrombocytopenia. Am J Hematol. 2004;77:358-62. https://doi.org/10.1002/ajh.20205
- Oka S, Ono K, Nohgawa M. Prediction of response to first-line therapy with ITP by flow cytometric analysis of bone marrow lymphocyte phenotypes. Int J Hematol. 2020;111:771-8. https://doi.org/10.1007/s12185-020-02847-4
- Arnason JE, Campigotto F, Neuberg D, Bussel JB. Abnormalities in IgA and IgM are associated with treatment-resistant ITP. Blood. 2012;119:5016-20. https://doi.org/10.1182/blood-2011-09-381020
- Aref S, El-Ghonemy MS, El-Aziz SA, Abouzeid T, Talaab M, El-Sabbagh A. Impact of serum immunoglobulins level and IL-18 promoter gene polymorphism among Egyptian patients with idiopathic thrombocytopenic purpura. Hematology. 2017;22:99-104. https://doi.org/10.1080/10245332.2016.1221213
- Audia S, Mahévas M, Samson M, Godeau B, Bonnotte B. Pathogenesis of immune thrombocytopenia. Autoimmun Rev. 2017;16:620-32. https://doi.org/10.1016/j.autrev.2017.04.012
- Kühne T. Diagnosis and management of immune thrombocytopenia in childhood. Hamostaseologie. 2017;37:36-44. https://doi.org/10.5482/HAMO-16-06-0017
- Onisâi M, Vlădăreanu AM, Spînu A, Găman M, Bumbea H. Idiopathic thrombocytopenic purpura (ITP) - new era for an old disease. Rom J Intern Med. 2019;57:273-83. https://doi.org/10.2478/rjim-2019-0014
- Perera M, Garrido T. Advances in the pathophysiology of primary immune thrombocytopenia. Hematology. 2017;22:41-53. https://doi.org/10.1080/10245332.2016.1219497
- George JN, Saucerman S. Platelet IgG, IgA, IgM, and albumin: correlation of platelet and plasma concentrations in normal subjects and in patients with ITP or dysproteinemia. Blood. 1988;72:362-5.
- Aboul-Fotoh LEM, Abdel Raheem MM, El-Deen MAB, Osman AMM. Role of CD4+CD25+ T cells in children with idiopathic thrombocytopenic purpura. J Pediatr Hematol Oncol. 2011;33:81-5. https://doi.org/10.1097/MPH.0b013e3181f46b82
- Nomura S. Advances in diagnosis and treatments for immune thrombocytopenia. Clin Med Insights Blood Disord. 2016;9:15-22. https://doi.org/10.4137/CMBD.S39643
- Arandi N, Mirshafiey A, Jeddi-Tehrani M, et al. Alteration in frequency and function of CD4⁺CD25⁺FOXP3⁺ regulatory T cells in patients with immune thrombocytopenic purpura. Iran J Allergy Asthma Immunol. 2014;13:85-92.
- Goubran H, Hart C, Othman I, Seghatchian J. Flow cytometry and immune thrombocytopenic purpura. Transfus Apher Sci. 2018;57:800-3. https://doi.org/10.1016/j.transci.2018.10.018
- Talaat RM, Elmaghraby AM, Barakat SS, El-Shahat M. Alterations in immune cell subsets and their cytokine secretion profile in childhood idiopathic thrombocytopenic purpura (ITP). Clin Exp Immunol. 2014;176:291-300. https://doi.org/10.1111/cei.12279
- Zhao Z, Yang L, Yang G, et al. Contributions of T lymphocyte abnormalities to therapeutic outcomes in newly diagnosed patients with immune thrombocytopenia. PLoS One. 2015;10:e0126601. https://doi.org/10.1371/journal.pone.0126601
Copyright and license
Copyright © 2026 The author(s). This is an open-access article published by Aydın Pediatric Society under the terms of the Creative Commons Attribution License (CC BY) which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.














