Abstract
Background: Pediatric autoimmune diseases (ADs) are more and more known as complicated conditions influenced by overlapping genetic and environmental factors; however, the coexistence of secondary ADs and their familial accumulation has not yet been explored thoroughly in pediatric populations. In this study we aimed to evaluate the occurrence and characteristics of secondary ADs among pediatric patients with autoimmune diseases followed in our pediatric rheumatology department and to examine the impact of a family history of autoimmunity on the development of secondary ADs.
Methods: We retrospectively reviewed the records of 488 pediatric patients followed in our pediatric rheumatology department who were diagnosed with autoimmune diseases. We have collected clinical, serological, and familial data. Secondary ADs were defined as newly diagnosed autoimmune diseases evolving after the initial diagnosis. Kaplan-Meier analysis and logistic regression were used to determine predictive factors.
Results: Secondary ADs were detected in 7% of patients. Systemic lupus erythematosus (SLE) (3.1%) was the most frequent, followed by autoimmune thyroid disease (0.8%), psoriasis (0.6%), and inflammatory bowel disease (0.6%). Among patients who developed SLE as a secondary diagnosis, the most frequent primary conditions were autoimmune hepatitis and immune thrombocytopenic purpura. Autoimmune hepatitis showed the strongest link with secondary AD development (OR=95.15, 95% CI: 19.07–474.70, p < 0.001). Patients with a positive family history of autoimmune diseases (FHADs) had a significantly higher likelihood of developing secondary autoimmune diseases (OR=5.11, 95% CI: 1.55–16.86, p=0.007). Meanwhile, we have seen that the probability of developing secondary ADs increased over time, reaching 26.6% over 15 years.
Conclusion: In this cohort, systemic lupus erythematosus was the most frequent secondary autoimmune disease, while secondary autoimmunity overall was more strongly associated with autoimmune hepatitis and a positive family history.
Keywords: children, autoimmunity, systemic lupus erythematosus, familial autoimmunity, autoimmune hepatitis
INTRODUCTION
Autoimmune diseases (ADs) are chronic disorders resulting from a breakdown of immunological tolerance to self-antigens and may affect either a single organ or multiple systems.1 Despite their heterogeneity, ADs often present with overlapping clinical features such as arthralgia, arthritis, Raynaud’s phenomenon, or fatigue. They are also frequently associated with various autoantibodies, as well as elevated proinflammatory cytokines such as TNF, IL-1, IL-6, IL-10, and IL-17.2 From a pathophysiological perspective, immune-mediated tissue injury is driven by dysregulated B- and T-cell responses, accompanied by infiltration of macrophages, neutrophils, and dendritic cells. Subsets of T helper cells, especially Th1 and Th17 cells, have been associated with intensifying the inflammatory cascade, whereas regulatory T cells play an essential role in maintaining tolerance. The defects of regulatory tools, together with altered cytokine signaling, are vital to the initiation and perpetuation of ADs.1-3
In the development of autoimmunity, environmental triggers have long been implicated. In particular, infectious agents play a central role, with Epstein–Barr virus and cytomegalovirus consistently linked to several autoimmune conditions.4 Genetic predisposition is another major factor in disease vulnerability. As evidenced by twin and family studies, both common and disease-specific genetic factors contribute to autoimmunity.1
The most common ADs in children include juvenile idiopathic arthritis (JIA), type 1 diabetes mellitus (DM), autoimmune thyroiditis, inflammatory bowel disease, and celiac disease.5,6 The global prevalence of ADs in children is anticipated at 5%, although it varies across regions.3,5 These diseases are causing chronic morbidity, decreased quality of life, and increased healthcare costs, with some cases leading to high mortality rates.7 Familial clustering of ADs is well known and suggests a genetic predisposition.8-12 Studies have shown that relatives of patients with autoimmune diseases such as rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), or JIA have high rates of autoimmune conditions, indicating a shared genetic and environmental factor.10,11,13
This study aims to characterize the co-occurrence of secondary ADs and explore the role of familial autoimmunity among pediatric patients followed in our department.
MATERIALS AND METHODS
Study design and population
This retrospective cohort study was conducted at the Pediatric Rheumatology Department of Hacettepe University, Ankara, and included 488 pediatric patients diagnosed with autoimmune diseases between 2005 and 2024. All patients were under 18 years of age at the time of initial diagnosis and had at least 6 months of clinical follow up. This study has been approved by the Hacettepe University Ethics Committee (approval date 24.04.2025, number 25/286) and conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from the legal guardians of all patients.
Definitions
The diagnoses of ADs were established based on validated international classification criteria. JIA was diagnosed using the International League of Associations for Rheumatology (ILAR) criteria.14 SLE was classified according to both the 2012 Systemic Lupus International Collaborating Clinics (SLICC) criteria and 2019 EULAR/ACR classification criteria.15,16 Autoimmune hepatitis (AIH) was diagnosed based on the International Autoimmune Hepatitis Group (IAIHG) criteria and confirmed by liver biopsy findings.17 Autoimmune thyroid disease (AITD) was diagnosed in the presence of elevated anti-thyroglobulin (TgA) and/or anti-thyroid peroxidase antibodies (TPOA), along with abnormal thyroid function tests. Celiac disease (CD) was diagnosed through positive anti-tissue transglutaminase (tTG) IgA and confirmed by small bowel biopsy. Inflammatory bowel disease (IBD) was confirmed by endoscopic and histopathological findings in patients with compatible clinical symptoms.
Other autoimmune conditions were defined according to the following established criteria: Juvenile Dermatomyositis (JDM) by the Bohan and Peter criteria and the 2017 EULAR/ACR classification criteria; Systemic Sclerosis (SS) by the 2013 ACR/EULAR classification criteria; and Sjögren’s syndrome by the 2016 ACR/EULAR criteria.18-21 Mixed connective tissue disease (MCTD) was diagnosed according to the Alarcón-Segovia criteria.22 Vitiligo and psoriasis were diagnosed by dermatologists based on characteristic clinical findings, with histopathological confirmation when necessary. Immune thrombocytopenic purpura (ITP) was diagnosed using the 2009 International Working Group (IWG) criteria, defined as a platelet count below 100,000/mm³ in the absence of other causes of thrombocytopenia.23 The diagnosis was made clinically based on exclusion, without reliance on disease-specific laboratory tests.
Secondary ADs were defined as a newly diagnosed autoimmune condition occurring after the initial diagnosis of a primary autoimmune disease, based on clinical findings, serological markers, and relevant classification criteria. The interval between primary and secondary diagnoses was recorded in years.
Family history of ADs was obtained from caregivers during clinical follow-up. Only first-degree relatives were considered. Reported conditions included autoimmune thyroid disease, rheumatoid arthritis, ankylosing spondylitis, psoriasis, celiac disease, juvenile idiopathic arthritis, type 1 diabetes mellitus, vitiligo, alopecia areata, inflammatory bowel disease, systemic lupus erythematosus, and cutaneous lupus erythematosus.
Statistical analysis
To summarize the study data, descriptive statistics were used. Continuous variables are presented as mean ± standard deviation (SD) when normally distributed, and as median (25th–75th percentile) when non-normally distributed. Categorical variables are presented as frequencies and percentages. Normality was assessed using the Shapiro–Wilk test and visual inspection of histograms and Q–Q plots. Group comparisons were managed using the chi-square test or Fisher’s exact test for categorical variables, and Student’s t-test or Mann–Whitney U test for continuous variables, whichever was appropriate. Logistic regression analysis was used to evaluate associations between clinical variables and the development of secondary autoimmune diseases (SAIDs), results expressed as odds ratios (ORs) and 95% confidence intervals (CIs). We used Kaplan–Meier analysis to estimate the cumulative probability of SAID development over time. A two-sided p-value < 0.05 was considered statistically significant.
RESULTS
This cohort involved 488 pediatric patients (353 females) diagnosed with several ADs. The mean age at primary AD onset was 8.89 ± 4.95 years. The median disease duration was 5.42 years (25th–75th percentile: 2.3–9.5). The most common AD in the cohort was JIA, which affected 213 patients (43.6%), with the most prevalent subtype being oligoarticular JIA (n=127), followed by polyarticular JIA (n=41).
Thirty-four patients (7%) had secondary ADs. The most frequent condition was SLE (n=15), followed by AITD, psoriasis, and IBD. Table 1 presents the detailed distribution of both primary and secondary autoimmune diseases observed in our cohort. The median disease duration was significantly longer in patients with secondary ADs than in those without (p=0.040). The median time between the diagnosis of the primary disease and the development of secondary ADs was 3.20 years (25th–75th percentile: 0.7–7.13). Since JIA was the most frequent AD in our cohort, no significant difference was found in the development of secondary autoimmunity concerning the diagnosis of oligoarticular JIA (p=0.250). Secondary autoimmunity was not reported in patients diagnosed with enthesitis-related arthritis (ERA) and psoriatic arthritis. Regarding specific primary diagnoses, a statistically significant difference was observed in patients with autoimmune hepatitis (p < 0.001), where 20.6% developed secondary ADs. Psoriasis and Sjögren’s Syndrome were less frequently associated with secondary ADs in both groups (p=0.450 and p=0.560, respectively) (Table 2).
| Table 1. Distribution of primary and secondary autoimmune diseases among pediatric patients (n=488) | ||
|---|---|---|
| Autoimmune Disease |
n (%) |
|
| JIA: juvenile idiopathic arthritis | ||
| Oligoarticular JIA |
|
|
| Polyarticular JIA |
|
|
| Enthesitis-related JIA |
|
|
| Psoriatic arthritis |
|
|
| Juvenile Dermatomyositis |
|
|
| Systemic lupus erythematosus |
|
|
| Scleroderma |
|
|
| Sjögren’s syndrome |
|
|
| Mixed connective tissue disease |
|
|
| Autoimmune hepatitis |
|
|
| Autoimmune thyroid disease |
|
|
| Inflammatory bowel disease |
|
|
| Psoriasis |
|
|
| Immune thrombocytopenic purpura |
|
|
| Type 1 diabetes mellitus |
|
|
| Vitiligo |
|
|
| AD: Autoimmune disease; JDM: Juvenile dermatomyositis | |||
| Table 2. Comparison of primary autoimmune diagnoses and clinical characteristics in patients with and without secondary autoimmune diseases | |||
| Characteristics |
|
|
|
| Female, n (%) |
|
|
|
| Median age at onset (years) |
|
|
|
| Median disease duration (years) |
|
|
|
| Systemic lupus erythematosus (%) |
|
|
|
| Juvenile idiopathic arthritis (%) |
|
|
|
| Scleroderma (%) |
|
|
|
| JDM (%) |
|
|
|
| Psoriasis (%) |
|
|
|
| Sjögren’s syndrome (%) |
|
|
|
| Autoimmune hepatitis (%) |
|
|
|
| Mixed connective tissue disease (%) |
|
|
|
| Immune thrombocytopenic purpura (%) |
|
|
|
| Autoimmune thyroid disease (%) |
|
|
|
In patients with a primary diagnosis of IBD, the secondary diagnosis was ERA. Among the 5 patients diagnosed with ITP, 2 developed SLE as a secondary diagnosis. Of the 10 patients with a primary diagnosis of autoimmune hepatitis, 7 developed secondary autoimmune diseases, including 6 cases of SLE and one case of Sjögren’s syndrome. In the 3 patients with a primary diagnosis of autoimmune thyroid diseases, secondary diseases included one case of SLE, a case of polyarticular JIA, and a case of psoriasis.
In our cohort, there were also cases of two autoimmune diseases coexisting at the time of diagnosis. One patient with a primary diagnosis of diabetes mellitus simultaneously developed autoimmune thyroid disease and enthesitis-related arthritis. In contrast, another patient with juvenile dermatomyositis was found to have concomitant diabetes mellitus and celiac disease as secondary autoimmune conditions. Among those who ended up developing a second autoimmune condition, three patients were diagnosed with psoriasis. Psoriasis appeared in three children. Two of them were on anti-TNF drugs at the time. The third one had not used any biologics before. Also, one child with ERA ended up developing ulcerative colitis while being treated with infliximab. A total of 249 of the 349 patients (71.5%) were positive for antinuclear antibodies (ANA). The homogeneous pattern was the most frequent among ANA-positive patients, occurring in 28.3% of cases. This was followed by the granular pattern (25.5%) and the dense fine speckled pattern (6.28%). A full breakdown of the remaining autoantibody findings is presented in Table 3.
| Table 3. Laboratory autoantibody and immunological test results in pediatric autoimmune diseases | ||
|---|---|---|
| Test |
|
|
| ANA: antinuclear antibody; dsDNA: double-stranded DNA; ANCA: anti-neutrophil cytoplasmic antibody; HLA: human leukocyte antigen; TPOA: anti-thyroid peroxidase antibody; TgA: anti-thyroglobulin antibody. | ||
| ANA |
|
|
| Anti-dsDNA |
|
|
| ANCA |
|
|
| HLA-B27 |
|
|
| Direct Coombs test |
|
|
| TPOA |
|
|
| TgA |
|
|
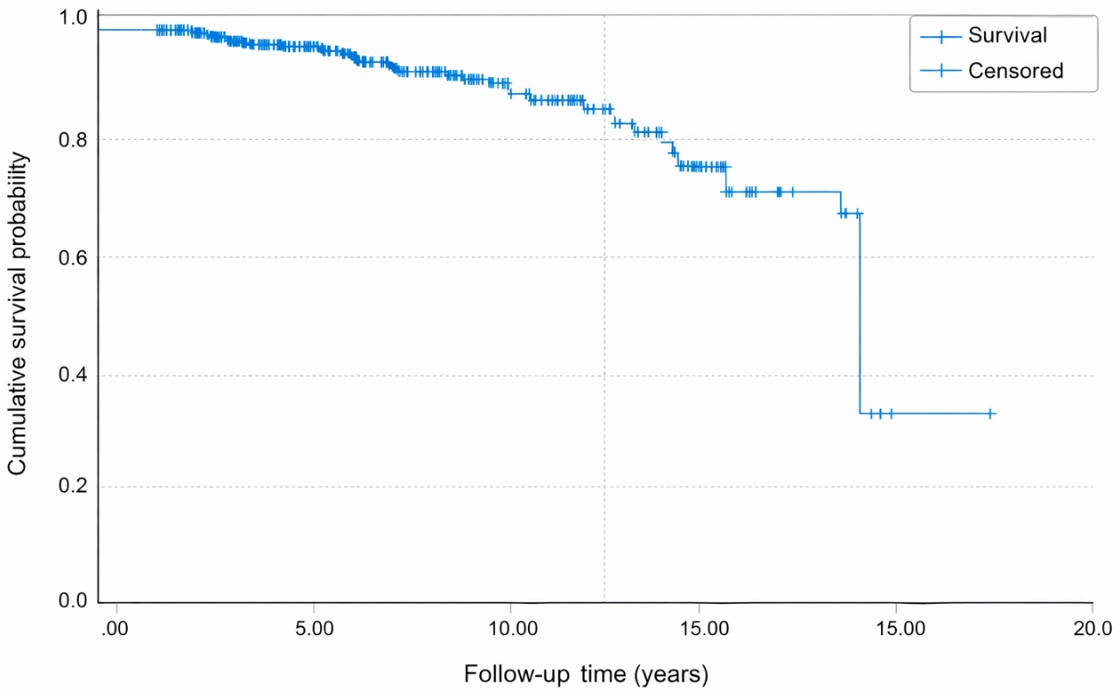
After univariate analyses, a logistic regression analysis revealed that certain primary autoimmune diseases significantly increased the likelihood of developing secondary autoimmune conditions. Primary diagnosis of autoimmune hepatitis showed the strongest association with the development of secondary autoimmune diseases (OR=95.15, 95% CI: 19.07–474.70, p < 0.001). Similarly, a positive family history of autoimmune diseases was significantly associated with the presence of secondary autoimmune conditions (OR=5.11, 95% CI: 1.55–16.86, p=0.007). The Kaplan-Meier estimates of the cumulative probability of developing a secondary autoimmune disease were 3.2% at 5 years (95% CI: 0.7%–5.7%), 9.6% at 10 years (95% CI: 2.8%–16.4%), and 26.6% at 15 years (95% CI: 2.4%–50.8%) (Figure 1).
Patients with a family history of autoimmune diseases have developed higher rates of secondary ADs compared to those without (13.8% versus 5.9%) (p=0.030). Among the cohort, 65 patients (13.3%) had at least one first-degree relative with a family history of autoimmune diseases (FHADs). The analysis revealed three main clusters of familial ADs in our cohort. AITD (4.9%), rheumatoid arthritis (RA) (2.1%), and DM (1.8%) were the most frequent autoimmune conditions found in the families of the patients. Table 4 presents the frequency of ADs observed in the families of pediatric patients.
| Table 4. Frequency of autoimmune diseases among families of pediatric patients (n=488) | ||
|---|---|---|
| The "Other*" category includes the following diseases, each with a frequency of 1 case (0.2%): Sarcoidosis, alopecia areata, cutaneous lupus erythematosus, celiac disease, and juvenile idiopathic arthritis. In 3 families, more than two diagnoses were present. Percentages are calculated based on the total study population (n=488). More than one autoimmune disease could be reported within the same family; therefore, percentages do not sum to 100% | ||
| Autoimmune Disease |
|
|
| Autoimmune thyroid disease |
|
|
| Rheumatoid arthritis |
|
|
| Diabetes mellitus |
|
|
| Ankylosing spondylitis |
|
|
| Psoriasis |
|
|
| Systemic lupus erythematosus |
|
|
| Ulcerative colitis |
|
|
| Psoriatic arthritis |
|
|
| Other* |
|
|
The family history of ADs was further analyzed. The results indicated that 30 patients (6.1%) had a family history of autoimmunity in the mother, 19 patients (3.9%) had it in the father, 8 patients (1.6%) had a sibling with AD, and 8 patients (1.6%) had both parents and siblings affected by autoimmunity. Table 5 presents the clinical characteristics and disease features of pediatric patients with and without FHADs.
|
FHAD: family history of autoimmune diseases Percentages are calculated within each column (n=65 for patients with FHADs and n=423 for patients without FHADs). |
|||
| Table 5. Comparison of clinical characteristics in patients with and without a reported family history of autoimmunity | |||
| Characteristic |
|
|
|
| Female, n (%) |
|
|
|
| Median age at onset (years) |
|
|
|
| Median disease duration (years) |
|
|
|
| Secondary autoimmune disease rate (%) |
|
|
|
| Systemic lupus erythematosus (%) |
|
|
|
| Juvenile idiopathic arthritis (%) |
|
|
|
| Scleroderma (%) |
|
|
|
| Psoriasis (%) |
|
|
|
| Sjögren’s syndrome (%) |
|
|
|
| Autoimmune hepatitis (%) |
|
|
|
| Mixed connective tissue disease (%) |
|
|
|
| Immune thrombocytopenic purpura (%) |
|
|
|
| Autoimmune thyroid disease (%) |
|
|
|
DISCUSSION
Autoimmune burden in juvenile idiopathic arthritis (JIA) was previously evaluated. But the occurrence of secondary autoimmune diseases across varied pediatric autoimmune conditions has not been clearly examined. This study gives a comprehensive assessment of secondary autoimmune diseases in this context and also insight into the role of familial autoimmunity.
We already know that in some cases, distinguishing between a new autoimmune diagnosis and a manifestation of the primary disease may be challenging. To minimize this potential misclassification, we required that all secondary diagnoses meet established international classification criteria and that satisfactory follow-up confirm their persistence as independent disease entities. Yet the possibility remains that some cases classified as secondary ADs may represent disease evolution rather than distinct comorbid autoimmunity, which we admit as a limitation of our study.
In our cohort of 488 pediatric patients, 7% developed secondary ADs. Systemic lupus erythematosus was the most frequent one (3.1%), followed by AITD, psoriasis, and IBD (each 0.6%). Juvenile idiopathic arthritis being the most common primary diagnosis, secondary AD development was not significantly associated with oligoarticular JIA. This finding differs from the study by Tronconi et al., which reported a 15.2% secondary AD rate in JIA, primarily AITD (10.1%).24 Our cohort showed a low prevalence of DM and celiac disease, but previous studies propose that clinicians should remain vigilant for additional autoimmune manifestations in patients with underlying endocrinopathies.25
Psoriasis was rarely a primary diagnosis with secondary ADs, but it was identified secondarily in three patients—two of whom had received anti-TNF therapy. This supports previous reports of psoriasis as a paradoxical effect of TNF inhibitors.26 However, one patient developed psoriasis without prior biologic treatment, and we considered that psoriasis itself may increase the likelihood of additional autoimmune diseases. It is reasonable that both mechanisms coexist— specifically, paradoxical drug-induced lesions and true secondary autoimmunity. Wu et al. showed that psoriasis increases the likelihood of further autoimmune diseases, highlighting the importance of close monitoring.27
Inflammatory bowel disease (IBD) was identified as a secondary AD in three patients. None of them had an ERA, and only one had received anti-TNF therapy. This contrasts with van Straalen et al., who found that ERA and a family history of autoimmunity significantly increased IBD risk in JIA.28 Our findings imply that IBD in non-ERA patients may involve distinct mechanisms.
Autoimmune hepatitis (AIH) showed the strongest association with secondary AD development (20.6%; p < 0.001), particularly with SLE and Sjögren’s syndrome as secondary diagnoses. This finding supports the previous studies that note the link and overlap between AIH and other autoimmune disorders. A retrospective study conducted on patients diagnosed with Sjögren’s syndrome found that 1.7% of patients with primary Sjögren’s syndrome had AIH.29 Given its high secondary ADs risk, patients with AIH need careful follow-up. Haslak et al. reported that ANA positivity in children rarely progressed to autoimmune disease, underscoring the need for reasonable interpretation in clinical practice.30 The co-occurrence of multiple autoimmune diseases in pediatric patients may reflect a complex relationship of genetic susceptibility, shared immunopathogenic pathways, and environmental triggers. Familial autoimmunity in our cohort supports the role of common inherited risk factors, particularly within HLA and non-HLA loci, in predisposing to polyautoimmunity.24
Problems in how B and T cells behave, along with the failure of both central and peripheral immune tolerance, seem to play a role in the development of multiple autoimmune conditions.3 Ongoing stimulations by infections may also contribute. Altogether, these overlapping mechanisms suggest that coexisting autoimmune diseases do not occur in isolation—they are likely part of a broader, shared disease process.
Familial autoimmunity was another key risk factor for secondary ADs development. Patients with a positive family history had a significantly higher secondary ADs rate. The most common familial autoimmune conditions were AITD (4.9%), rheumatoid arthritis (2.1%), and diabetes mellitus (1.8%). Though JIA patients with a family history of autoimmunity have been reported to show clustering of AITD, RA, and psoriasis, our findings showed no significant difference in SAID development between familial and non-familial JIA cases.24 Similar patterns were observed in scleroderma and Sjögren’s syndrome. Notably, while previous studies indicated familial aggregation in juvenile dermatomyositis (JDM), our JDM patients had no family history of autoimmunity, highlighting possible regional or population-specific differences.31
In our own group, ITP was seen in 14.7% of children with a family history of autoimmunity. Interestingly, two out of five of those ITP cases later developed SLE as a second autoimmune disease. This relatively high rate of disease progression is important from a clinical point of view and is consistent with earlier studies suggesting that some children with ITP may later develop SLE. In a national French cohort, 22% of ANA-positive children with autoimmune cytopenia ended up being diagnosed with SLE, which made up 4.4% of the whole AIC group.32 These findings highlight why it’s important to follow pediatric ITP patients closely over time, especially those with unusual antibody results or a family history of autoimmune conditions.
Our study has important strengths, including data from a relatively large pediatric cohort, systematic evaluation of a broad spectrum of autoimmune diseases (not just JIA), and a long observation period. Additionally, the combination of detailed family history data provides valuable insight into the genetic and environmental background of pediatric autoimmunity. On the other hand, we admit certain limitations. The retrospective, single-center design may limit the generalizability of our findings. Some disease subgroups have relatively small sample sizes, which may reduce power to detect significant associations, and reliance on caregiver reports of family history may introduce recall bias. Therefore, caution should be kept when generalizing these frequencies to the general pediatric population.
In our large pediatric cohort, 7% of patients developed secondary autoimmune diseases, most commonly systemic lupus erythematosus. Autoimmune hepatitis and a positive family history emerged as key risk factors for additional autoimmunity. These findings highlight the need for long-term follow-up and careful monitoring of children with autoimmune diseases, particularly those with these risk factors.
Ethical approval
This study has been approved by the Hacettepe University Ethics Committee (approval date 24.04.2025, number 25/286). Written informed consent was obtained from the participants.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Anaya JM. Common mechanisms of autoimmune diseases (the autoimmune tautology). Autoimmun Rev. 2012;11:781-4. https://doi.org/10.1016/j.autrev.2012.02.002
- Pisetsky DS. Pathogenesis of autoimmune disease. Nat Rev Nephrol. 2023;19:509-24. https://doi.org/10.1038/s41581-023-00720-1
- Wang L, Wang FS, Gershwin ME. Human autoimmune diseases: a comprehensive update. J Intern Med. 2015;278:369-95. https://doi.org/10.1111/joim.12395
- Barzilai O, Sherer Y, Ram M, Izhaky D, Anaya JM, Shoenfeld Y. Epstein-Barr virus and cytomegalovirus in autoimmune diseases: are they truly notorious? A preliminary report. Ann N Y Acad Sci. 2007;1108:567-77. https://doi.org/10.1196/annals.1422.059
- Głowińska-Olszewska B, Szabłowski M, Panas P, et al. Increasing co-occurrence of additional autoimmune disorders at diabetes type 1 onset among children and adolescents diagnosed in years 2010-2018-single-center study. Front Endocrinol (Lausanne). 2020;11:476. https://doi.org/10.3389/fendo.2020.00476
- Müller KE, Lakatos PL, Arató A, et al. Incidence, Paris classification, and follow-up in a nationwide incident cohort of pediatric patients with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2013;57:576-82. https://doi.org/10.1097/MPG.0b013e31829f7d8c
- Somers EC, Thomas SL, Smeeth L, Hall AJ. Are individuals with an autoimmune disease at higher risk of a second autoimmune disorder? Am J Epidemiol. 2009;169:749-55. https://doi.org/10.1093/aje/kwn408
- Ansell BM, Bywaters EG, Lawrence JS. Familial aggregation and twin studies in Still’s disease. Juvenile chronic polyarthritis. Rheumatology. 1969;2:37-61.
- Rossen RD, Brewer EJ, Sharp RM, et al. Familial rheumatoid arthritis: a kindred identified through a proband with seronegative juvenile arthritis includes members with seropositive, adult-onset disease. Hum Immunol. 1982;4:183-96. https://doi.org/10.1016/0198-8859(82)90034-9
- van Straalen JW, de Roock S, Giancane G, et al. Prevalence of familial autoimmune diseases in juvenile idiopathic arthritis: results from the international Pharmachild registry. Pediatr Rheumatol Online J. 2022;20:103. https://doi.org/10.1186/s12969-022-00762-y
- Shao YHJ, Chen YM. Parental autoimmunity and offspring risks of rheumatic diseases: a nationwide population-based study. Rheumatology (Oxford). 2024;63:2189-98. https://doi.org/10.1093/rheumatology/kead562
- Al-Mayouf SM, Alrasheedi A, Almsellati I, et al. Familial aggregation of juvenile idiopathic arthritis with other autoimmune diseases: impact on clinical characteristics, disease activity status and disease damage. Int J Rheum Dis. 2021;24:1080-5. https://doi.org/10.1111/1756-185X.14167
- Richard-Miceli C, Criswell LA. Emerging patterns of genetic overlap across autoimmune disorders. Genome Med. 2012;4:6. https://doi.org/10.1186/gm305
- Petty RE, Southwood TR, Manners P, et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol. 2004;31:390-2.
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64:2677-86. https://doi.org/10.1002/art.34473
- Aringer M, Costenbader K, Daikh D, et al. 2019 European league against rheumatism/American College of rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71:1400-12. https://doi.org/10.1002/art.40930
- Hennes EM, Zeniya M, Czaja AJ, et al. Simplified criteria for the diagnosis of autoimmune hepatitis. Hepatology. 2008;48:169-76. https://doi.org/10.1002/hep.22322
- Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med. 1975;292:344-7. https://doi.org/10.1056/NEJM197502132920706
- Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med. 1975;292:403-7. https://doi.org/10.1056/NEJM197502202920807
- van den Hoogen F, Khanna D, Fransen J, et al. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013;65:2737-47. https://doi.org/10.1002/art.38098
- Shiboski CH, Shiboski SC, Seror R, et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Ann Rheum Dis. 2017;76:9-16. https://doi.org/10.1136/annrheumdis-2016-210571
- Alarcon-Segovia D, Villareal M. Classification and diagnostic criteria for mixed connective tissue disease. In: Kasukawa R, Sharps GC, editors. Mixed connective tissue diseases and anti- nuclear antibodies. Amsterdam: Elsevier; 1987: 33e40.
- Rodeghiero F, Stasi R, Gernsheimer T, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113:2386-93. https://doi.org/10.1182/blood-2008-07-162503
- Tronconi E, Miniaci A, Pession A. The autoimmune burden in juvenile idiopathic arthritis. Ital J Pediatr. 2017;43:56. https://doi.org/10.1186/s13052-017-0373-9
- Alpigiani MG, Cerboni M, Bertini I, et al. Endocrine autoimmunity in young patients with juvenile chronic arthritis. Clin Exp Rheumatol. 2002;20:565-8.
- Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40:233-40. https://doi.org/10.1016/j.semarthrit.2010.04.003
- Wu JJ, Nguyen TU, Poon KYT, Herrinton LJ. The association of psoriasis with autoimmune diseases. J Am Acad Dermatol. 2012;67:924-30. https://doi.org/10.1016/j.jaad.2012.04.039
- van Straalen JW, Krol RM, Giancane G, et al. Increased incidence of inflammatory bowel disease on etanercept in juvenile idiopathic arthritis regardless of concomitant methotrexate use. Rheumatology (Oxford). 2022;61:2104-12. https://doi.org/10.1093/rheumatology/keab678
- Karp JK, Akpek EK, Anders RA. Autoimmune hepatitis in patients with primary Sjögren’s syndrome: a series of two-hundred and two patients. Int J Clin Exp Pathol. 2010;3:582-6.
- Haslak F, Yildiz M, Altun I, et al. Anti-nuclear antibody testing in children: how much is really necessary? Pediatr Int. 2021;63:1020-5. https://doi.org/10.1111/ped.14592
- Niewold TB, Wu SC, Smith M, Morgan GA, Pachman LM. Familial aggregation of autoimmune disease in juvenile dermatomyositis. Pediatrics. 2011;127:e1239-46. https://doi.org/10.1542/peds.2010-3022
- Granel J, Fernandes H, Bader-Meunier B, et al. Antinuclear antibody-associated autoimmune cytopenia in childhood is a risk factor for systemic lupus erythematosus. Blood. 2024;143:1576-85. https://doi.org/10.1182/blood.2023021884
Copyright and license
Copyright © 2026 The author(s). This is an open-access article published by Aydın Pediatric Society under the terms of the Creative Commons Attribution License (CC BY) which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.