Abstract
Objectives: Biologic disease-modifying antirheumatic drugs (bDMARDs) have been frequently used to treat juvenile idiopathic arthritis (JIA) resistant to classical (c) DMARDs. There is concern that vaccines may reduce vaccine effectiveness due to their immunosuppressive effects. This study aimed to evaluate the prevalence of anti-hepatitis B surface (HBs) antibody positivity in JIA patients treated with bDMARDs and to compare it with that in JIA patients treated with cDMARDs and in healthy controls.
Materials and Methods: Anti-HBs antibody positivity and titers were compared between patients with JIA treated with bDMARDs or only cDMARDs, followed in our clinic, and healthy controls aged 2-20 years. All participants were vaccinated in infancy according to the routine vaccination schedule in our country. Anti-HBs titers ≥10 IU/L were considered seroprotective.
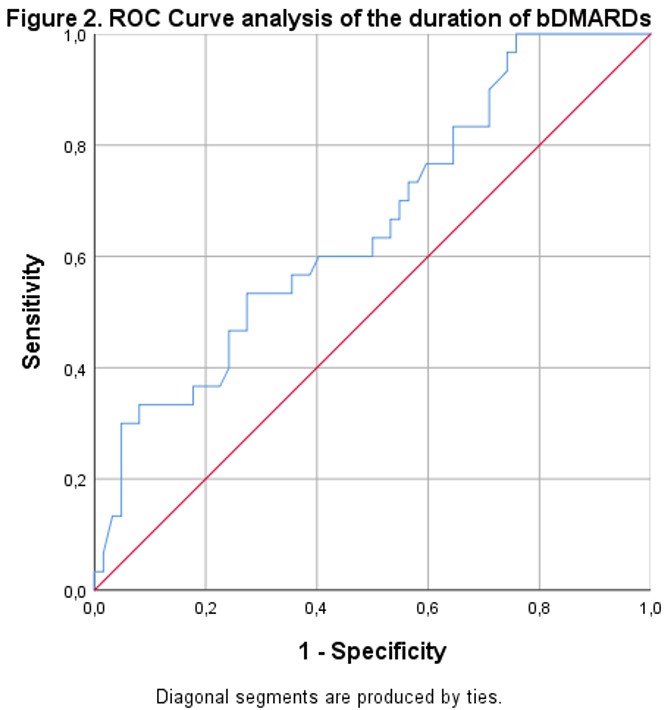
Results: Ninety-two JIA patients receiving cDMARDs, 92 receiving bDMARDs, and 91 healthy controls were included in the study. The median time from the last vaccination to the study was 12.5 (min-max:3.5-17.55) years in controls, 12.5 (min-max:1.75-19.5) years in patients with cDMARDs, and 13.0 (min-max:2.25-19.0) years in patients with bDMARDs (p=0.060). Anti-HBs was positive in 50 (54.9%) controls, 66 (71.7%) patients with cDMARDs, and 62 (67.4%) patients with bDMARDs (p=0.048). Age, time from vaccination to study, and duration of bDMARD use were found to be risk factors for anti-HBs negativity in univariate regression analyses. According to the multivariable analysis of these three variables, the duration of bDMARD use was an independent risk factor for anti-HBs negativity (OR: 1.023, 95% CI: 1.005-1.041, p=0.013). Anti-HBs negativity was associated with a duration of bDMARDs longer than 32 months (AUC: 0.658, 95%CI: 0.541-0.776, p=0.014) with 60.0% sensitivity, and 59.7% specificity.
Conclusions: A longer duration of bDMARDs was found to be a risk factor for anti-HBs negativity. Physicians should be careful in terms of anti-HBs negativity when the duration of biologics use is prolonged.
Keywords: Anti-HBs antibody, bDMARDs, biologic drugs, hepatitis B, juvenile idiopathic arthritis, tumor necrosis factor inhibitors
INTRODUCTION
Juvenile idiopathic arthritis (JIA) is the most common cause of chronic arthritis in children. Immunosuppressants and biologics are the cornerstones of treatment, but both raise concerns about reducing the protective effects of vaccines.1-3 Studies evaluating the effects of classical disease-modifying anti-rheumatic drugs (cDMARDs) and biological (b) DMARDs on vaccine responses in JIA patients are controversial.4 It is known that not only bDMARDs and cDMARDs, but also the disease itself can reduce the immunogenicity of vaccines.5,6
In recent decades, neonatal immunization programs have been implemented in many countries, but hepatitis B virus (HBV) infection remains a major public health problem.7 Hepatitis B vaccine has been included in the national immunization programs for all children and at-risk groups. It has been routinely administered in Turkey as three doses at birth, 1 month, and 6 months since 1998. The immunization program has significantly decreased the seroprevalence of hepatitis B surface antigen (HBsAg).8
The American College of Rheumatology (ACR) recommended antibody testing for HBV prior to initiating methotrexate or tumor necrosis factor α inhibitors (TNFi) for only JIA patients at risk of HBV infection.9 The authors identified patients born in geographic regions with HBsAg prevalence ≥2% as a risk factor for HBV infection.9 The prevalence of HBsAg positivity was determined 4.0% in our country, so our JIA patients are at risk for HBV infection.10
The European League Against Rheumatism (EULAR) and the Paediatric Rheumatology European Society (PRES) have recommended adhering to national immunization guidelines for inactivated vaccines, such as the HBV vaccine, in JIA patients receiving cDMARDs or bDMARDs.1,2 Low seroprotective antibody titers have also been reported in JIA patients.11 However, the immunization and treatment guidelines do not specify whether booster vaccination or routine screening for protective antibody levels is required in JIA patients after initiation of treatment.1,2,9 Over the past decade, there has been increasing evidence that inactivated vaccines, including HBV, do not exacerbate the underlying disease or cause serious side effects, and studies have shown that the immunogenicity of the HBV vaccine is adequate.2,3,12 In contrast, several studies have reported that bDMARDs such as TNFi reduce the immunogenicity of the HBV vaccine.13,14
Our study aimed to evaluate HBV vaccination responses in JIA patients treated with cDMARDs and bDMARDs and to compare them with those in healthy controls. We also investigated whether the use of bDMARDs influences seroprotective antibody levels in JIA patients.
MATERIALS AND METHODS
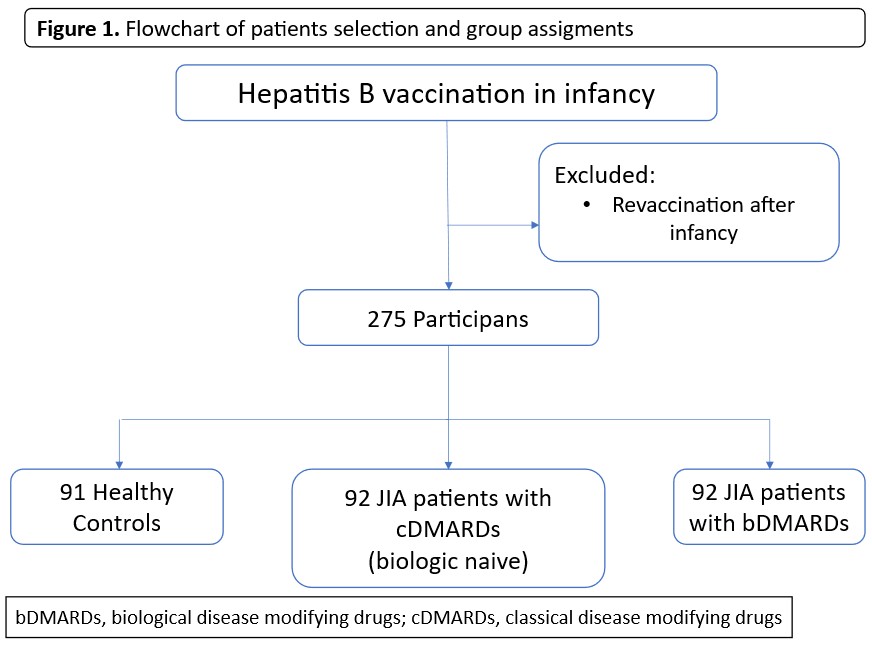
Our study was carried out between July 2018 and July 2019, and included 92 JIA patients treated with cDMARDs, 92 patients treated with bDMARDs, and 91 healthy controls aged 2-20 years. The flowchart of patients and group assignments is shown in Figure 1. All JIA patients who were followed for at least three months and attended the outpatient clinic during this period were included in the study. The control group consisted of age and sex-matched healthy children who applied to general pediatrics clinics for routine examination. Patients or healthy children who received one or more booster doses of HBV vaccine after infancy were excluded. All patients and controls were vaccinated with the hepatitis B vaccine in infancy (0, 1, and 6 months schedule). Vaccination schedules were confirmed from vaccination cards. The diagnosis of JIA was made according to the International League of Associations for Rheumatology (ILAR) classification criteria.15 Demographic data, clinical characteristics, and treatment durations were obtained from the medical records of JIA patients. The Childhood Health Assessment Questionnaire (CHAQ) and the Juvenile Arthritis Disease Activity Score 27 (JADAS27) were recorded for patients at the last visit.
JIA patients were divided into two groups: patients treated with cDMARDs and with bDMARDs. Ninety-two JIA patients received standard therapies such as nonsteroidal anti-inflammatory drugs, systemic or intra-articular steroids, and cDMARDs (methotrexate, sulfasalazine, leflunomide, cyclosporine, hydroxychloroquine), and did not receive any bDMARDs treatment throughout the course of their disease. Ninety-two JIA patients received bDMARDs (anti-TNFα, anti-interleukin [IL]-6, anti-IL-1, or abatacept [cytotoxic T lymphocyte-associated antigen-4 immunoglobulin]) with/without other standard therapies and cDMARDs.
HBsAg, hepatitis B surface antibody (anti-HBs), and hepatitis B core antibody (anti-HBc) concentrations were measured using electrochemiluminescence immunoassay (ECLIA) (Cobas 8000, Roche Diagnostics, Germany). Anti-HBs titers ≥10 IU/L were considered seroprotective.16 Patients who had an anti-HBs titer ≤10 IU/L were considered anti-HBs negative.
This study was carried out in accordance with the Declaration of Helsinki. Informed consent was obtained from all parents and patients, and from controls older than 18 years of age. This study was approved by the institutional ethics committee (2018/223)
Statistical analysis
Statistical analyses were performed using IBM SPSS 20 (SPSS, Inc., Chicago, IL). Categorical variables are presented as frequencies and percentages. The distribution of continuous variables was evaluated with the Kolmogorov-Smirnov test. Continuous variables are shown as mean and standard deviation (SD) if normally distributed, and median and minimum-maximum if abnormally distributed. The Mann-Whitney U test was used to compare two groups, and the Kruskal-Wallis test was used to compare three groups of continuous variables that were abnormally distributed. Logistic regression analysis was performed to investigate which factors were associated with anti-HBs negativity. The variables found to be significant in the univariate logistic regression analysis were subjected to multivariate regression analysis to determine the independent risk factors for anti-HBs negativity. The time interval between the last HBV vaccination and antibody testing varied among participants. We accounted for its potential effect by including the time from vaccination to the study as a covariate in a multivariate logistic regression model. Model calibration was evaluated using the Hosmer–Lemeshow goodness-of-fit test. Receiver operating characteristic (ROC) analysis was used to determine the optimal cutoff point for the duration of bDMARD treatment. A p-value of <0.05 was considered significant.
RESULTS
Ninety-two patients received bDMARDs, 92 patients received cDMARDs, and 91 healthy controls were included in the study. The median age was 13.0 (4.0-18.0) years in controls, 13.0 (2.25-20.0) years in cDMARDs group, and 13.5 (2.75-19.5) years in bDMARDs group (p=0.059). Forty-five (49.5%) of controls, 57 (62.0%) of patients treated with cDMARDs, and 58 (63.0%) of patients treated with bDMARDs were female (p=0.117). The median time from the last vaccination to the study was 12.5 (3.5-17.55) years in controls, 12.5 (1.75-19.5) years in patients with cDMARDs, and 13.0 (2.25-19.0) years in patients with bDMARDs (p=0.060).
Anti-HBs was positive in 50 (54.9%) controls, 66 (71.7%) patients with cDMARDs, and 62 (67.4%) patients with bDMARDs (p=0.048) (Table 1). The omnibus chi-square test indicated a significant association between the variables. However, Bonferroni-corrected post-hoc pairwise comparisons (three 2×2 sub-analyses) revealed no statistically significant differences between any specific category pairs (all p-values > 0.0167). This suggests that the overall chi-square significance reflects a distributed pattern across cells rather than a discrete pairwise difference. Anti-HBs titers were not different among the three groups (p=0.100). None of the 275 participants had serological evidence of active or previous hepatitis B infection. HBsAg and anti-HBc were negative in all participants.
| Values are summarized using median (min-max) and n (%). DMARDs, disease modifying anti-rheumatic drugs | ||||
| Table 1. Comparison of the characteristics of healthy controls, patients with juvenile idiopathic arthritis treated with classical disease-modifying drugs, and with biological disease-modifying drugs | ||||
| Variable | Control (n=91) | Patients with cDMARDs (n=92) |
Patients with bDMARDs (n=92) |
p value |
| Age, year | 13.0 (4.0-18.0) | 13.0 (2.25-20.0) | 13.5 (2.75-19.5) | 0.059 |
| Gender | 0.117 | |||
| Female | 45 (49.5) | 57 (62.0) | 58 (63.0) | |
| Male | 46 (50.5) | 35 (38.0) | 34 (37.0) | |
| Time from last vaccination to the study, years | 12.5 (3.5-17.55) | 12.5 (1.75-19.5) | 13.0 (2.25-19.0) | 0.060 |
| Anti-HBs | 0.048 | |||
| Positive (≥10 IU/L) | 50 (54.9) | 66 (71.7) | 62 (67.4) | |
| Negative (<10 IU/L) | 41 (45.1) | 26 (28.3) | 30 (32.6) | |
| Anti-HBs titer (IU/L) | 12.95 (2-1000) | 19.45 (2-1000) | 26.68 (2-1000) | 0.100 |
Demographic, clinical, and laboratory features of the patients are shown in Table 2. The median age at the study was 13.0 (2.25-20.0) years in the cDMARDs group and 13.5 (2.75-19.5) years in the bDMARDs group (p=0.018). The median age at diagnosis was 9.4 (1.33-16.0) years in the cDMARDs group and 8.38 (1.42-16.0) years in the bDMARDs group (p=0.481).
|
Values are summarized using median (min-max) and n (%). ANA: anti-nuclear antibody; CHAQ: childhood health assessment questionnaire; DMARDs: disease modifying anti-rheumatic drugs; ERA: enthesitis related arthritis; HLA, human leukocyte antigen; JADAS, juvenile arthritis disease activity score; JIA: juvenile idiopathic arthritis; RF: rheumatoid factor. #Systemic methylprednisolone *Fisher’s exact test |
||||
| Table 2. Comparison of the characteristics of patients with juvenile idiopathic arthritis treated with classical disease modifying drugs and with biological disease modifying drugs | ||||
| Variable |
|
|
|
|
| Age, year |
|
|
|
|
| Gender |
|
|||
| Female |
|
|
|
|
| Male |
|
|
|
|
| Age at diagnosis, years |
|
|
|
|
| Time from last vaccination to the study, years |
|
|
|
|
| Follow up, months |
|
|
|
|
| JIA subtypes |
|
|||
| Oligoarticular JIA |
|
|
|
|
| ERA |
|
|
|
|
| RF- polyarticular JIA |
|
|
|
|
| RF+ polyarticular JIA |
|
|
|
|
| Systemic JIA |
|
|
|
|
| Psoriatic arthritis |
|
|
|
|
| Undifferentiated |
|
|
|
|
| ANA positivity (169/184) |
|
|
|
|
| HLA-B27 positivity (108/184) |
|
|
|
|
| Total number of involved joints in disease course |
|
|
|
|
| Patients received steroids# |
|
|
|
|
| Duration of steroids#, months |
|
|
|
|
| Cumulative steroid# dosage, grams |
|
|
|
|
| Patients received cDMARDs |
|
|
|
|
| Duration of cDMARDs, months |
|
|
|
|
| Duration of bDMARDs, months |
|
|
|
|
| Active arthritis at last visit |
|
|
|
|
| CHAQ score at last visit |
|
|
|
|
| JADAS27 at last visit |
|
|
|
|
| Steroid# at last visit |
|
|
|
|
| DMARDs at last visit |
|
|
|
|
| Biologics at last visit |
|
|
|
|
The time from vaccination to the study was 13.0 (2.75-19.0) versus 12.5 (1.75-19.5), and the follow-up time was 49.0 (5-172) versus 29.5 (3-132), respectively, in the bDMARDs group compared with the cDMARDs group, p=0.018, p<0.001, respectively. Oligoarticular JIA patients constituted the majority of patients in both patient groups. JIA patient subtypes are shown in Table 2.
The number of involved joints, the number of patients who received steroids, and the total duration and dosage of steroid treatment were greater in the bDMARDs group (p<0.001). The median duration of cDMARD use was longer in the bDMARDs group (p < 0.001). The disease activity scores at the last visit were not different between cDMARDs and bDMARDs groups (p>0.05) (Table 2).
Sixty-three (68.5%) patients used only one bDMARD throughout the disease course, and 29 (31.5%) patients used more than one bDMARD due to drug switching because of treatment failure. During the course of the disease, 61 patients in the bDMARD group received etanercept, 32 received adalimumab, 16 received tocilizumab, 14 received infliximab, 5 received anakinra, 3 received canakinumab, and 2 patients received abatacept. Because the number of patients receiving non-TNFi biologics was very small, subgroup sizes were highly unbalanced. Therefore, no inferential statistical comparison between bDMARD subclasses was performed.
Patients treated with cDMARDs and bDMARDs were evaluated separately for anti-HBs positivity (Table 3). The age at diagnosis was higher in anti-HBs negative patients with cDMARDs [10.6 (3.83-15.33) versus 8.08 (1.33-16.0), p=0.033]. Other demographic, clinical, and laboratory features were not different between anti-HBs-positive and anti-HBs-negative patients in the cDMARDs group (p>0.05) (Table 3). The number of joints involved was higher [6 (1-26) versus 3 (1-30) joints, p=0.007], and the duration of treatment with bDMARDs was longer [38.0 (7.5-115) versus 29.42 (1.33-100) months, p=0.014] in anti-HBs negative patients treated with bDMARDs.
|
Values are summarized using median (min-max) and n (%). ANA, anti-nuclear antibody; CHAQ, childhood health assessment questionnaire; DMARDs, disease modifying anti-rheumatic drugs; ERA, enthesitis related arthritis; HLA, human leukocyte antigen; JADAS, juvenile arthritis disease activity score; JIA, juvenile idiopathic arthritis; RF, rheumatoid factor #Systemic methylprednisolone |
||||||
| Table 3. Comparison of anti-HBs positive and negative patients with juvenile idiopathic arthritis treated with and without biologics | ||||||
| Variable |
|
|
||||
|
|
|
|
|
|
|
|
| Age, years |
|
|
|
|
|
|
| Gender |
|
|
||||
| Female |
|
|
|
|
||
| Male |
|
|
|
|
||
| Age at diagnosis, years |
|
|
|
|
|
|
| Time from vaccine to the study, years |
|
|
|
|
|
|
| Follow up, months |
|
|
|
|
|
|
| JIA subtypes |
|
|
||||
| Oligoarticular JIA |
|
|
|
|
||
| ERA |
|
|
|
|
||
| RF- polyarticular JIA |
|
|
|
|
||
| RF+ polyarticular JIA |
|
|
|
|
||
| Systemic JIA |
|
|
|
|
||
| Psoriatic arthritis |
|
|
|
|
||
| Undifferentiated |
|
|
|
|
||
| ANA positivity (84/85) |
|
|
|
|
|
|
| HLA B27 positivity (51/57) |
|
|
|
|
|
|
| Total number of involved joints in disease course |
|
|
|
|
|
|
| Patients received steroids# |
|
|
|
|
|
|
| Duration of steroids#, months |
|
|
|
|
|
|
| Cumulative steroid# dosage, g |
|
|
|
|
|
|
| Patients receiving DMARDs |
|
|
|
|
|
|
| Duration of DMARDs, months |
|
|
|
|
|
|
| Duration of biological drugs, months |
|
|
|
|
|
|
| Active arthritis at last visit |
|
|
|
|
|
|
| CHAQ score at last visit |
|
|
|
|
|
|
| JADAS27 at last visit |
|
|
|
|
|
|
| Steroids# at last visit |
|
|
|
|
|
|
| DMARDs at last visit |
|
|
|
|
|
|
| Biologics at last visit |
|
|
|
|
|
|
Age, time from vaccination to study, and duration of bDMARD use were found to be risk factors for anti-HBs negativity in univariate regression analyses (Table 4). According to the multivariable analysis of these three variables, the duration of bDMARDs was an independent risk factor for anti-HBs negativity (OR: 1.023, 95% CI: 1.005-1.041, p=0.013) (Table 4). Goodness-of-fit was assessed using the Hosmer–Lemeshow test, which indicated an adequate model fit (χ² = 10.586, p = 0.226).
|
ANA, anti-nuclear antibody; CHAQ, childhood health assessment questionnaire; DMARDs, disease modifying anti-rheumatic drugs; ERA, enthesitis related arthritis; HLA, human leukocyte antigen; JADAS, juvenile arthritis disease activity score; JIA, juvenile idiopathic arthritis; RF, rheumatoid factor *All JIA patients were included in univariate and multivariate logistic regression analyses $Age, time from vaccine to the study and duration of bDMARDs therapy variables were included to the multivariate logistic regression analysis #Systemic methylprednisolone |
||||
| Table 4. Factors affecting the anti-HBs negativity in patients with juvenile idiopathic arthritis (n=184)* | ||||
| Variables |
|
|
||
|
|
|
|
|
|
| Age, years |
|
|
|
|
| Gender |
|
|
||
| Age at diagnosis, years |
|
|
||
| Duration of disease, months |
|
|
||
| Time from vaccine to the study, years |
|
|
|
|
| Number of involved joints |
|
|
||
| ANA positivity |
|
|
||
| HLA B27 positivity |
|
|
||
| Receiving steroid# therapy |
|
|
||
| Duration of steroid# therapy, months |
|
|
||
| Cumulative total steroid# dosage (grams) |
|
|
||
| Receiving cDMARD therapy |
|
|
||
| Duration of cDMARD therapy, months |
|
|
||
| Receiving bDMARDs therapy |
|
|
||
| Duration of bDMARDs therapy, months |
|
|
|
|
| Active arthritis at last visit |
|
|
||
| CHAQ score at last visit |
|
|
||
| JADAS27 at last visit |
|
|
||
| Steroid# treatment at last visit |
|
|
||
| cDMARDs treatment at last visit |
|
|
||
| bDMARDs treatment at last visit |
|
|
||
ROC analysis was used to evaluate the optimal cutoff for the duration of bDMARD treatment for anti-HBs negativity. Anti-HBs negativity was associated with a duration of bDMARDs treatment longer than 32 months (AUC: 0.658, 95%CI: 0.541-0.776, p=0.014) with 60.0% sensitivity, and 59.7% specificity (Figure 2).
DISCUSSION
Our study showed that the use of bDMARDs is not a risk factor for anti-HBs negativity in JIA patients, whereas longer bDMARD use is a significant risk factor for decreased seroprotective antibody levels. Therefore, we believe that patients on bDMARDs should be followed more closely for anti-HBs negativity, especially if the duration of treatment is prolonged.
Previous studies have shown that anti-HBs positivity rates ranging 60.9-69.2% in healthy children, and the percentage of seroprotective anti-HBs levels decreases over time in healthy children vaccinated in infancy.8,17-21 Anti-HBs positivity rate in healthy children was 54.9% in our study. Consistent with the literature, our study also showed that anti-HBs positivity decreases with age. However, interestingly, the percentage of anti-HBs-negative individuals was higher in the control group than in the JIA groups in our study. The lack of significant Bonferroni-adjusted pairwise differences implies that the observed association in the overall chi-square test may result from small, non-specific deviations across multiple cells rather than a strong difference between any two individual categories. In a previous study, Heijstek et al.5 showed that patients with JIA had lower levels of mumps, rubella, diphtheria, and tetanus-specific antibodies, except for measles, compared to healthy controls. Interestingly, they found higher antibody levels in patients with active disease.5 They could also not explain the reason, but they concluded that nonspecific immune activation during active disease plays a role in modulating antibody concentrations.5 The immune activation in the disease course may have caused the anti-HBs positivity to be higher in our JIA patients than in controls. When we compared anti-HBs-negative and anti-HBs-positive patients with JIA, we could not find any difference in disease activity scores.
We found that anti-HBs titers did not differ between healthy controls, JIA patients on cDMARDs, and bDMARDs. Szczygielska et al. investigated the anti-HBs concentration in 56 JIA patients vaccinated in infancy and who had received bDMARDs for at least 3 months; 22 (39.3%) were anti-HBs negative, and 34 (60.7%) were seropositive.11 The rate of anti-HBs positivity was similar to that in our study. However, they did not include healthy controls for comparison with JIA patients. In addition, no statistically significant correlation was found between the anti-HBs concentration and the time after the last vaccination, or the current age of the patients.11
Anti-HBs positivity rate was not lower in JIA patients treated with cDMARDs or bDMARDs in our study. Maritsi et al.22 reported that anti-HBs antibody titers were lower in treatment-naïve JIA patients than in matched healthy children. Similarly, Çakmak et al. investigated HBV vaccine responses in treatment-naïve JIA patients and reported lower anti-HBs positivity in JIA patients than in healthy controls.6 These findings suggested that the disease itself reduces anti-HBs positivity. But we could not evaluate treatment-naïve JIA patients.
Salinas GF et al.14 evaluated HBV vaccine responses in SpA patients receiving only TNFi treatment, and none of these patients received concomitant immunosuppressive drugs. Serum samples were collected at baseline before HBV vaccination, and at 6, 10, 22, and 26 weeks after vaccination.14 The median antibody titers in patients treated with or without TNFi were 10 IU/ml (0-115 IU/ml) and 595 IU/ml (65-2190 IU/ml) at week 26, respectively (p=0.005).14 They claimed that there was a strong suppression of the induction of T-cell-dependent antibodies against HBV in patients receiving TNFi treatment.14 Their study evaluated anti-HBs levels at 6th months postvaccination, but we evaluated long-term antibody concentrations and did not find any significant difference in anti-HBs concentrations.
In our study, we did not find any differences in anti-HBs levels or positivity rates by JIA subtype. Kostik et al. investigated the risk factors for non-protective antibody levels against HBV, measles, mumps, rubella, and diphtheria. They reported that patients treated with bDMARDs had the lowest probability of having protective antibody levels against HBV, measles, mumps, and diphtheria compared to patients treated with MTX and NSAIDs.3 In addition, they found that the most important predictor affecting anti-HBs antibodies was systemic JIA subtype.3 Univariate logistic regression analysis showed that age, time from vaccination to the study, and duration of bDMARDs were statistically significant risk factors for anti-HBs negativity in our study. The time interval between the last HBV vaccination and antibody testing varied among participants. We included this variable in our multivariate regression model to avoid the potential misleading effect. We evaluated these three variables using multiple logistic regression analysis, and only the duration of bDMARDs was found to be an independent risk factor for anti-HBs negativity. We also compared anti-HBs-negative and anti-HBs-positive patients separately. We confirmed that the duration of bDMARD therapy was longer in anti-HBs-negative patients in the bDMARD group. The frequencies of memory B cells, antibody-secreting cells, and plasma cells associated with maintenance of anti-HBs antibody titers in healthy subjects.23 Anolik et al. have shown that TNFi treatment reduces the percentage of memory B cells in the peripheral blood.24 The reason why anti-HBs negativity was seen more frequently in our patients with a longer bDMARDs duration may be related to this aspect.
There are some limitations of our study. First, although the majority of bDMARDs received were TNFi, some patients received other bDMARDs. Second, we did not evaluate treatment-naïve JIA patients. Also, patients on bDMARDs may represent a subgroup with more severe disease, which could confound the results. But we included disease activity scores, the number of active joints, other treatments such as steroids and cDMARDs, and the duration of cDMARD use. These variables were not significant in univariate analysis. The strengths of our study are that it covers the long-term period after the vaccination and that the effects of both cDMARDs and bDMARDs on vaccine responses were also assessed.
CONCLUSION
To our knowledge, this is the first study to show a correlation between vaccine response and the duration of bDMARD use. The rate of anti-HBs positivity did not differ between patients treated with cDMARDs, bDMARDs, and healthy controls. However, the duration of biologic therapy seems to be a risk factor for anti-HBs negativity. Patients receiving bDMARDs should be carefully monitored for anti-HBs negativity, especially if the duration of treatment is prolonged. Prospective studies with larger, more homogeneous patient groups are needed.
Ethical approval
This study has been approved by the Erciyes University Clinical Researches Ethics Committee (approval date 18.04.2018, number: 2018/223). Informed consent was obtained from all parents and patients, and from controls older than 18 years.
Source of funding
This study was funded by Erciyes University Scientific Research Projects Coordination Section (Project number: TSA-2018-8357)
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Heijstek MW, Ott de Bruin LM, Bijl M, et al. EULAR recommendations for vaccination in paediatric patients with rheumatic diseases. Ann Rheum Dis. 2011;70:1704-12. https://doi.org/10.1136/ard.2011.150193
- Jansen MHA, Rondaan C, Legger GE, et al. EULAR/PRES recommendations for vaccination of paediatric patients with autoimmune inflammatory rheumatic diseases: update 2021. Ann Rheum Dis. 2023;82:35-47. https://doi.org/10.1136/annrheumdis-2022-222574
- Kostik MM, Lubimova NA, Fridman IV, Goleva OV, Kharit SM. The vaccine coverage and vaccine immunity status and risk factors of non-protective levels of antibodies against vaccines in children with juvenile idiopathic arthritis: cross-sectional Russian tertiary Centre study. Pediatr Rheumatol Online J. 2021;19:108. https://doi.org/10.1186/s12969-021-00594-2
- Friedman MA, Curtis JR, Winthrop KL. Impact of disease-modifying antirheumatic drugs on vaccine immunogenicity in patients with inflammatory rheumatic and musculoskeletal diseases. Ann Rheum Dis. 2021;80:1255-65. https://doi.org/10.1136/annrheumdis-2021-221244
- Heijstek MW, van Gageldonk PGM, Berbers GAM, Wulffraat NM. Differences in persistence of measles, mumps, rubella, diphtheria and tetanus antibodies between children with rheumatic disease and healthy controls: a retrospective cross-sectional study. Ann Rheum Dis. 2012;71:948-54. https://doi.org/10.1136/annrheumdis-2011-200637
- Çakmak F, Çakan M, Demir F, et al. Hepatitis B vaccination response of treatment-naive patients with juvenile idiopathic arthritis. Rheumatol Int. 2022;42:1199-205. https://doi.org/10.1007/s00296-021-04833-3
- Jeng WJ, Papatheodoridis GV, Lok ASF. Hepatitis B. Lancet. 2023;401:1039-52. https://doi.org/10.1016/S0140-6736(22)01468-4
- Karatekin G, Kilinç M, Gulcan Öksuz B, Iğde M. Hepatitis B seroprevalence in children and women and the impact of the hepatitis B vaccination program in the Black Sea Region of Turkey. J Infect Dev Ctries. 2013;7:960-5. https://doi.org/10.3855/jidc.4102
- Beukelman T, Patkar NM, Saag KG, et al. 2011 American College of Rheumatology recommendations for the treatment of juvenile idiopathic arthritis: initiation and safety monitoring of therapeutic agents for the treatment of arthritis and systemic features. Arthritis Care Res (Hoboken). 2011;63:465-82. https://doi.org/10.1002/acr.20460
- Tozun N, Ozdogan O, Cakaloglu Y, et al. Seroprevalence of hepatitis B and C virus infections and risk factors in Turkey: a fieldwork TURHEP study. Clin Microbiol Infect. 2015;21:1020-6. https://doi.org/10.1016/j.cmi.2015.06.028
- Szczygielska I, Hernik E, Gazda A, Kołodziejczyk B, Gietka P. Assessment of anti-HBs antibody concentration in children with juvenile idiopathic arthritis treated with biological drugs, vaccinated against viral type B hepatitis in infancy. Reumatologia. 2020;58:15-20. https://doi.org/10.5114/reum.2020.93508
- Nerome Y, Akaike H, Nonaka Y, et al. The safety and effectiveness of HBV vaccination in patients with juvenile idiopathic arthritis controlled by treatment. Mod Rheumatol. 2016;26:368-71. https://doi.org/10.3109/14397595.2015.1085608
- Haykir Solay A, Eser F. High dose hepatitis B vaccine is not effective in patients using immunomodulatory drugs: a pilot study. Hum Vaccin Immunother. 2019;15:1177-82. https://doi.org/10.1080/21645515.2019.1574151
- Salinas GF, De Rycke L, Barendregt B, et al. Anti-TNF treatment blocks the induction of T cell-dependent humoral responses. Ann Rheum Dis. 2013;72:1037-43. https://doi.org/10.1136/annrheumdis-2011-201270
- Petty RE, Southwood TR, Manners P, et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol. 2004;31:390-2.
- European Consensus Group on Hepatitis B Immunity. Are booster immunisations needed for lifelong hepatitis B immunity? Lancet. 2000;355:561-5. https://doi.org/10.1016/S0140-6736(99)07239-6
- He F, Ma YJ, Zhou TY, et al. The serum anti-HBs level among children who received routine hepatitis B vaccination during infancy in Mianyang City, China: a cross-sectional study. Viral Immunol. 2016;29:40-8. https://doi.org/10.1089/vim.2015.0073
- Yue X, Ge C, Zhuge S, et al. Changes and analysis of anti-HBs titres after primary immunization in 1- to 16-year-old Chinese children: a hospital-based study. J Viral Hepat. 2018;25:373-80. https://doi.org/10.1111/jvh.12818
- Bayhan GI, Balli SE, Demir H, Baydar Z. How does the immunogenicity of hepatitis B vaccine change over the years in childhood? Hum Vaccin Immunother. 2021;17:2768-72. https://doi.org/10.1080/21645515.2021.1902724
- Tan S, Li S, Dong J, et al. Evaluation of factors associated the expression of anti-HBs in children in Hunan Province, China. BMC Pediatr. 2022;22:697. https://doi.org/10.1186/s12887-022-03718-z
- Behre U, Bleckmann G, Crasta PD, et al. Long-term anti-HBs antibody persistence and immune memory in children and adolescents who received routine childhood hepatitis B vaccination. Hum Vaccin Immunother. 2012;8:813-8. https://doi.org/10.4161/hv.19898
- Maritsi D, Vartzelis G, Soldatou A, Garoufi A, Spyridis N. Markedly decreased antibody titers against hepatitis B in previously immunised children presenting with juvenile idiopathic arthritis. Clin Exp Rheumatol. 2013;31:969-73.
- Doi H, Yoshio S, Yoneyama K, et al. Immune determinants in the acquisition and maintenance of antibody to hepatitis B surface antigen in adults after first-time hepatitis B vaccination. Hepatol Commun. 2019;3:812-24. https://doi.org/10.1002/hep4.1357
- Anolik JH, Ravikumar R, Barnard J, et al. Cutting edge: anti-tumor necrosis factor therapy in rheumatoid arthritis inhibits memory B lymphocytes via effects on lymphoid germinal centers and follicular dendritic cell networks. J Immunol. 2008;180:688-92. https://doi.org/10.4049/jimmunol.180.2.688
Copyright and license
Copyright © 2026 The author(s). This is an open-access article published by Aydın Pediatric Society under the terms of the Creative Commons Attribution License (CC BY) which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.